|
|
JEV IgG Rapid Diagnostic Test Cassette,WB/S/P
|
Product Details:
Payment & Shipping Terms:
|
| Package: | Neutral | Box: | White |
|---|
JEV IgG Rapid Test Cassette,WB/S/P
Intended use
The product is used for in vitro characterization of human veins in whole blood, serum or plasma samples of encephalitis b virus immunoglobulin G (IgG) antibody.
Epidemic encephalitis b(encephalitis b):It is an acute infection caused by encephalitis b virus and transmitted by mosquitoes. The high mortality and disability rate of encephalitis b is one of the main infectious diseases that threaten the health of people, especially children. Fall for peak season, disease epidemic areas distribution is closely related to the distribution of mosquito, encephalitis b is high endemic areas in China, in the 1960 s and early 70 s national pandemic broke out after the 70 s as a wide range of encephalitis b vaccination, je incidence is markedly reduced, in recent years to maintain in low level. And now, the number of reported cases of encephalitis b in China is between 5,000 and 10,000 each year, but there are outbreaks or epidemics in some areas. Since mosquitoes can carry the virus through the winter and can be passed from egg to egg, they are not only vectors of transmission, but also long-term storage hosts. After the mosquito infected with je bites the human body, the virus first proliferates in local tissue cells and lymph nodes, as well as vascular endothelial cells, invading the blood stream and forming viremia. The disease depends on the number of viruses, virulence and the immune function of the body. The vast majority of infected people do not get sick and have hidden infection. When the amount of invasive virus is large, virulence is strong, and the immune function of the body is insufficient, then the virus continues to multiply and spread throughout the body through blood. Because the virus has neurophilic nature, it can break through the blood-brain barrier and enter the central nervous system. In clinic, it is used for the auxiliary diagnosis of patients with encephalitis b virus infection.
【Test principle】
The kit employs a combination of colloidal gold immunochromatography,the T line is pre-coated with recombinant encephalitis b virus antigen,and the C line is pre-coated with goat anti-mouse IgG antibody,the mouse anti human IgG monoclonal antibody coated on colloidal gold.The kit is used to qualitatively detect encephalitis b virus immunoglobulin G (JEV-IgG) antibody in human serum, plasma or whole blood. Testing positive samples, JEV-IgG antibody in the sample can be captured on anti human IgG monoclonal antibody of colloidal gold and will bind to the formation of immune complexes,by the chromatography effect complexes and sample in internal flow on nitrocellulose membrane.When the complexes through the T line with the recombinant of JEV antiagen will bind to form a burgundy colored with the "Au-(mouse anti human IgG monoclonal antibody)-(JEV-IgG)-(JEV-Ag)".The rest of the complexes through the C line with goat anti-mouse IgG antibody will bind to form a burgundy colored. Testing negative samples, the samples without JEV-IgG antibody will not bind to the formation of immune complexes ,so only the color in C line.
Principal component
1. Test card/bar: 1 person/card or bar * 20T, consists of sample mat, Colloidal gold pad, nitrocellulose membrane and absorbent paper.The test line is pre-coated with recombinant encephalitis b virus antigen and the control line is pre-coated with goat anti-mouse IgG antibody, the gold standard pad is pre-coated with colloidal gold labeled mouse anti human IgG monoclonal antibody.
2. Sample diluent : 1 bottle,20mM pH7.4 PBS buffer solution.
Storage conditions and term of validity
The unopened kit should be stored under 4-30℃ and under this condition the kit can be used until the expiry date labeled on the kit which normally lasts for 12 months from date of manufacture.
When humidity is less than 60%, it will be used within 1 hour and when humidity is more than 60%, it will be used immediately.
Sample requirements
The whole blood was venous blood.Serum samples were collected by vein in a conventional manner.The plasma samples can be treated with heparin, sodium citrate and EDTA. The samples may be stored refrigerated (2-8℃) for as long as 5days or at -20℃ or below for 3 monthes.Do not freeze and thaw sample more than once. Turbid or precipitated samples should be centrifuged or filtered for clarification before testing.
Assay procedure
Please read the instructions thoroughly before testing..
Allow the test cassette and the sample to reach room temperature before testing.
Make sure the lab’s humidity less than 60%(RH<60%) and keep the experiment temperature is 18-30℃.
In the case of the kit and the sample, the temperature of the lab is unreinstated, and when the temperature of the lab is unmet, it can't be operated on, in case the results of the experiment are affected.
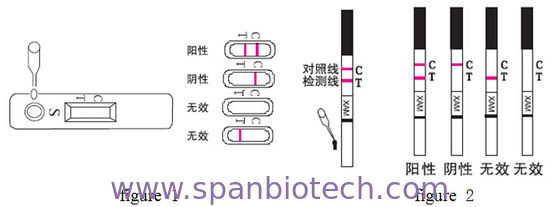
Test card detection method (figure 1)
1. Take the test card from the pouch.
2. Make sure that the test card is placed on flat surface during the testing.
3. Adding 10 µL serum, plasma samples or 20µL whole blood to be directly added to the add hole.Add the sample diluent 100 µL (about 2-3 drops).
4. The interpretation result within 15-20 minutes is invalid after 20 minutes.
figure 1 figure 2
Test bar detection method (figure 2)
1. Take the test bar from the pouch. Placed on flat surface during the testing.
2. Adding 10 µL serum, plasma samples or 20µL whole blood to be directly added to the add hole.Add the sample diluent 100 µL (about 2-3 drops).
3. The interpretation result within 15-20 minutes is invalid after 20 minutes.
Interpetation of assay result
1. Nagative result:Only a red line appears in the position of the control line (C).
2. Positive result:The control line (C) and the test line (T) position appear in two red strips.
3. Invalid results:The quality control line (C) has no red stripe
4. The invalid results should resume experiment again, when should be in strict accordance with the instructions operation, if the test result is invalid, its relationship with local suppliers or customer service with our company for technical consultation to determine whether products exist.
Limitations of the test method
1. A high concentration of nonspecific IgG antibodies, and the binding site of the antibody against the specific IgG antibodies will decrease the sensitivity of the test, which might weaken the test line.
2. The high concentration of the encephalitis IgM antibody will be associated with a specific IgG antibody, which will reduce the sensitivity of the test, and it could be a false negative result.
3. The reagent is qualitative and cannot be used to determine the antibody content
4. This reagent is used for the detection of whole blood,serum or plasma,not for saliva, urine or other bodily fluids.
5. Like all diagnostic reagents, the results must be combined with other clinical symptoms obtained by the physician
.
6. This product inspection result is only for clinical reference, should not serve as the only basis for clinical diagnosis and treatment.The clinical management of patients should be combined with its symptoms/signs, other medical history and laboratory examination, treatment response and epidemiological information such as the comprehensive consideration.
7. The interpretation of positive results should be further determined in combination with clinical symptoms and other etiological methods.
8. The negative results indicated that the IgG antibody of rubella virus was not detected, but if the IgG antibody content of rubella virus in the sample was lower than the minimum detectable limit of the kit, the negative result could also be obtained.
Product performance index
1. The positive coincidence rate, precision, negative coincidence rate and sensitivity of this product meet the quality standard requirements, and the product quality is stable within the validity period..
2. The results of cross - reaction and endogenous interference with other pathogens are as follows.:Hepatitis e virus IgG antibody,Hepatitis a virus IgG antibody,Hepatitis b virus surface antigen antibody,Hepatitis c virus IgG antibody,IgG antibody of treponema pallidum,HIV antibody,Mycoplasma pneumoniae IgG antibody,IgG antibody of chlamydia pneumoniae,Adenovirus IgG antibodies,IgG antibody of respiratory syncytial virus,Influenza a virus IgG antibody,Influenza b virus IgG antibodies,EV71 IgG antibody,Measles virus IgG antibody,Cytomegalovirus IgG antibody,The mumps virus IgG antibody,B encephalitis virus IgG antibody,Group A IgG antibody,Pertussis virus IgG antibody,The poliovirus IgG antibody,Rheumatoid factor,Antinuclear antibodies,Anti-mitochondrial antibody (AMA) positive samples,High concentrations of non-specific IgG (serum total IgG) and high concentrations of non-specific IgM (serum total IgM) do not interfere with this product.When the IgG antibody concentration of rubella virus is too high, the HOOK effect will appear. C line band is weakened, and it is suggested that the specimen should be diluted 10 times to be retested.The blood lipid content in the sample was higher than that of 6mmol/L, and the content of bilirubin was higher than 40, and the hemoglobin was greater than 5.0g /L, which had an effect on the experimental results.There was no significant difference between this rapid test and similar rapid test.With comparable public rapid test to clinical comparative study of 1000 cases of clinical specimens, the results of the rapid test and similar rapid tests total coincidence rate was 97.9%, sensitivity 94.8%, specificity 98.9%, the results of similar rapid tests and the rapid test was no significant difference.
Considerations
1. The positive samples obtained by the rapid test should be confirmed by other methods.
2. The rapid test should be sealed and kept in check.The test bar should be tested as soon as possible after being removed from the packaging, so as to avoid placing it in the air for too long, causing the damp.
3. The depth of the test line color is not necessarily associated with the titer of the antibody in the sample, and the results of the interpretation after 20 minutes are invalid.
4. When the antibody levels of the encephalitis b virus IgG in the sample is extremely high, the C line may decrease, which is a normal phenomenon.
5. All the waste samples and reagents should be treated as potential infectious agents.
Span Biotech Ltd.
Tel: +86(755)89589611
WhatsAPP:+8618823462100 (Wechat)
Web:www.spanbio.com
Contact Person: Ms. Anna Lee
Tel: +86-755-89589611
Fax: 86-755-89580096